 
Since there is more particle movement than within a solid, liquids have a higher kinetic energy value. This means that liquids have a fixed volume (no matter the shape of their container) as long as the temperature and pressure are held constant. However, the interaction is strong enough to keep the particles attracted to each other. Due to the weaker forces holding their particles together, liquids conform to the shape of their container. They are more loosely packed than a solid. In the liquid state, particles flow around each other. These properties include elasticity, conductivity, light transmittance, plasticity, and more. Due to their different force interactions, these categories of solids have different physical and chemical properties. These classes include metals, minerals, (glass) ceramics, organic molecules, composite materials, semiconductors, nanomaterials, and biomaterials. Whether or not we realize it, there are so many types of solids all around us-from table salt to a wooden chair! Since the types of forces and bonding between particles can vary, there are different classes of solids. Because their particles are so densely packed, solids tend to have a high density and are hard to compress further without the use of great external force. 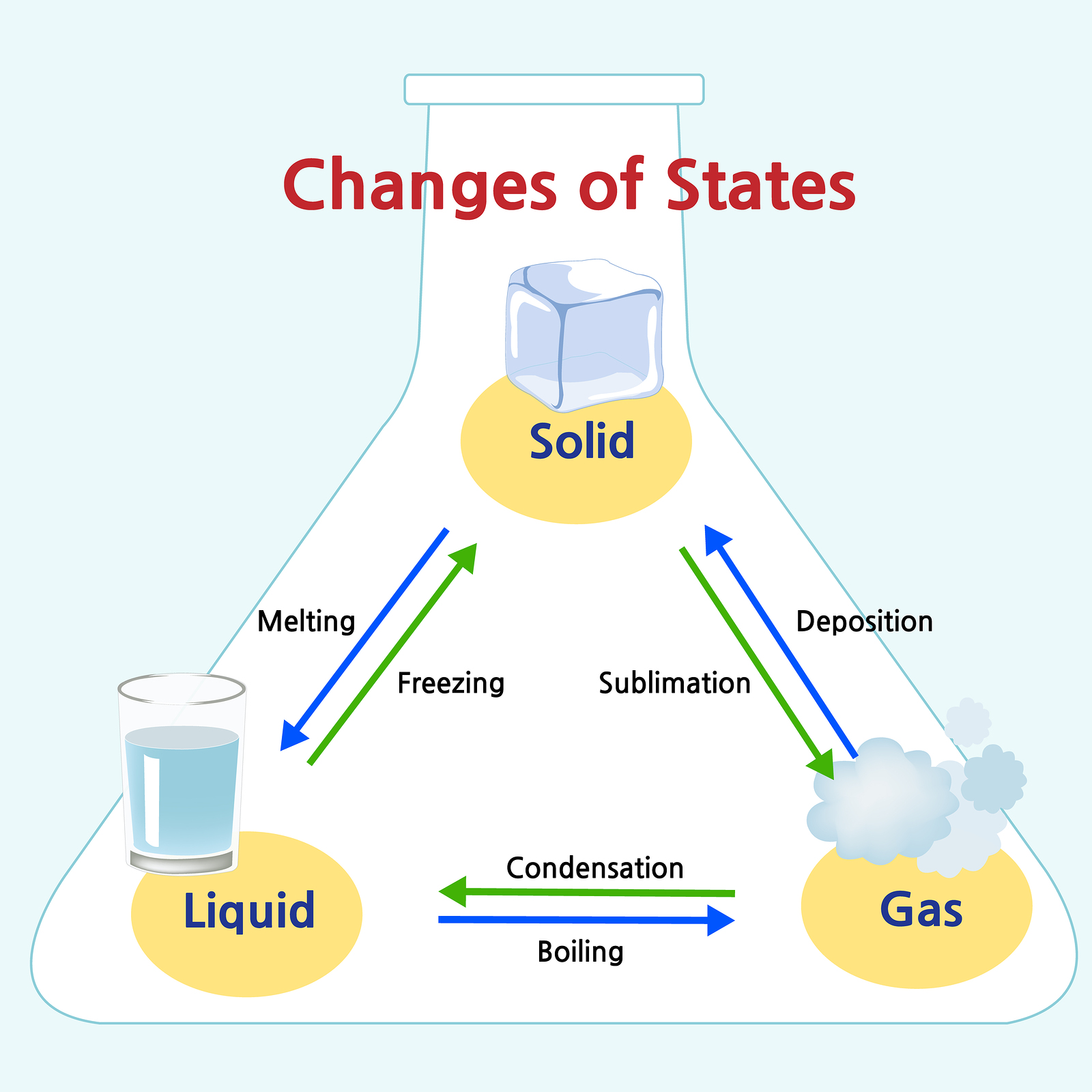
Solids have a definite shape and volume, meaning they have a fixed position and will not conform to the shape of their container. However, when there are extreme and rapid temperature changes, an indefinitely shaped solid will most likely be the result. Usually, when conditions are steady (such as slow and gradual cooling/heating), the particles have a chance to align uniformly. The materials present and the conditions in which a solid is created dictate whether it will be a crystalline or amorphous solid. The particles of the compound bind in either an organized, geometric lattice or a random, unstructured shape. As a result, solids have the lowest kinetic energy of all the states of matter. In other words, they stay in their fixed positions. Due to the strong forces holding them together, the particles of a solid are only able to move back and forth in small vibrations. In the solid state, particles tightly pack together in a fixed arrangement. Additionally, there are many intermediate states, many of which only exist under extreme conditions in total, there are twenty! Due to their difference in properties being their distinguishing factors, let’s go over some of the states of matter below: Solids There are four main states of matter if you include plasma. The properties of a substance, such as its density, conductivity, and viscosity, also vary depending on its state of matter. Solids have a fixed shape and volume, while liquids have a fixed volume but can take the shape of their container, and gases have neither a fixed shape nor a fixed volume. The behavior of matter also changes depending on its state. But if the water is heated to a high enough temperature, it will become a gas (steam), and if it is cooled to a low enough temperature, it will become a solid (ice). For example, at room temperature and pressure, water is a liquid. The state of a substance depends on its temperature and pressure. There are three main states of matter: solid, liquid, and gas. The states of matter refer to the physical forms that matter can take. Phase and Matter are occasionally used as synonyms however, it is possible to form several phases that are in the same state of matter (such as how solids can have different crystalline structures).For more in-depth information on the concept of matter, check out this article! Matter– anything that has mass and occupies space it constitutes atoms and compounds, which compose physical and chemical properties.A moving electric charge in a magnetic field experiences a force perpendicular to its velocity. Magnetic field – a region around a magnet or an electric current that describes the magnetic influence on moving electric charges, currents, and magnetic materials.Properties of Solids, Liquids, and Gases.In this tutorial, you will learn about the four main states of matter (solid, liquid, gas, and plasma), as well as some intermediate states of matter, by reading about their properties, applications, and examples. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
